 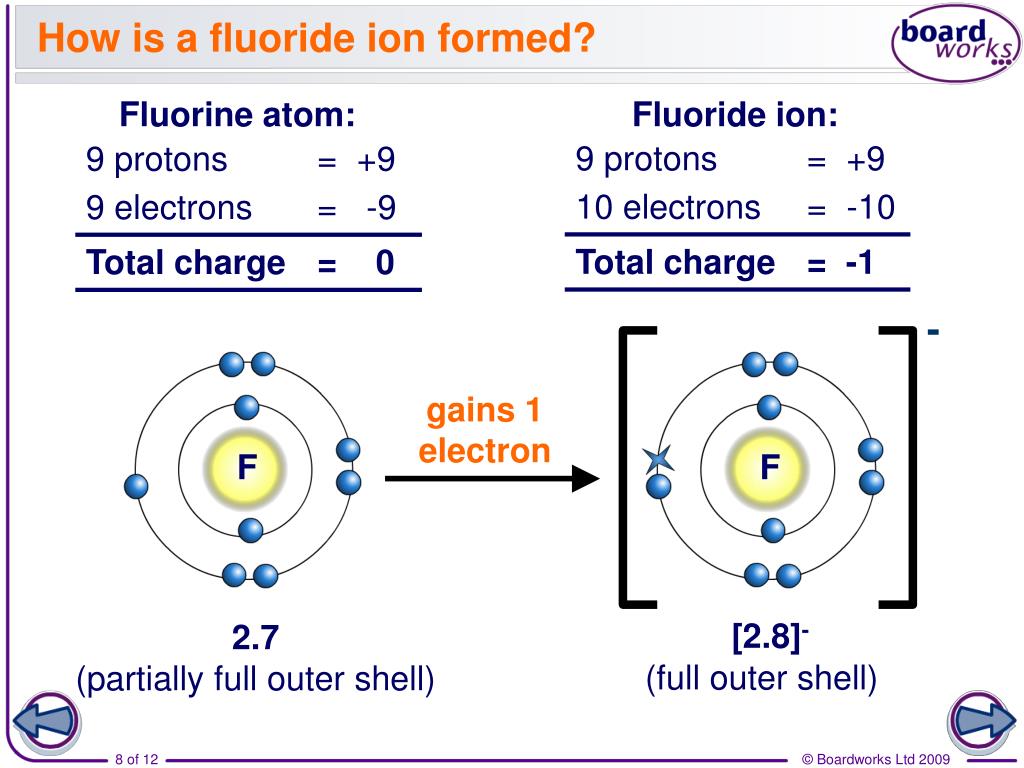 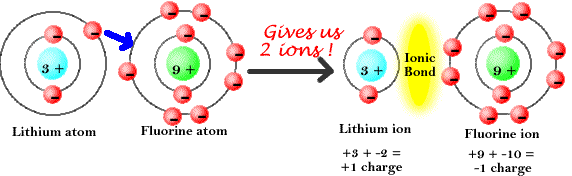
This page is shared under a CK-12 license and was authored, remixed, and/or curated by Melissa Alviar-Agnew, Henry Agnew, and Lance S. The atomic mass unit (amu) is a unit of mass equal to one-twelfth the mass of a carbon-12 atom.As a result, a neutral atom must have an equal number of protons and electrons. The positive charge on a proton is equal in magnitude to the negative charge on an electron.Protons and neutrons have approximately the same mass, but they are both much more massive than electrons (approximately 2,000 times as massive as an electron).Like protons, neutrons are bound into the atom's nucleus as a result of the strong nuclear force. Neutrons are a type of subatomic particle with no charge (they are neutral).An anion has a net negative electrical charge, which means it has more electrons than protons. A cation has a net positive electrical charge, which means it has more protons than electrons. There are two types of ions: cations and anions. Protons are bound together in an atom's nucleus as a result of the strong nuclear force. Ions are identified by a superscript that shows the sign and size of the electric charge for example Ca +2. Protons are a type of subatomic particle with a positive charge.Electrons are a type of subatomic particle with a negative charge.b) Develop a relationship (in the form of a single sentence or. Negative ions have protons than electrons. \): Properties of Subatomic Particles Particle Positive ions have protons than electrons. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
